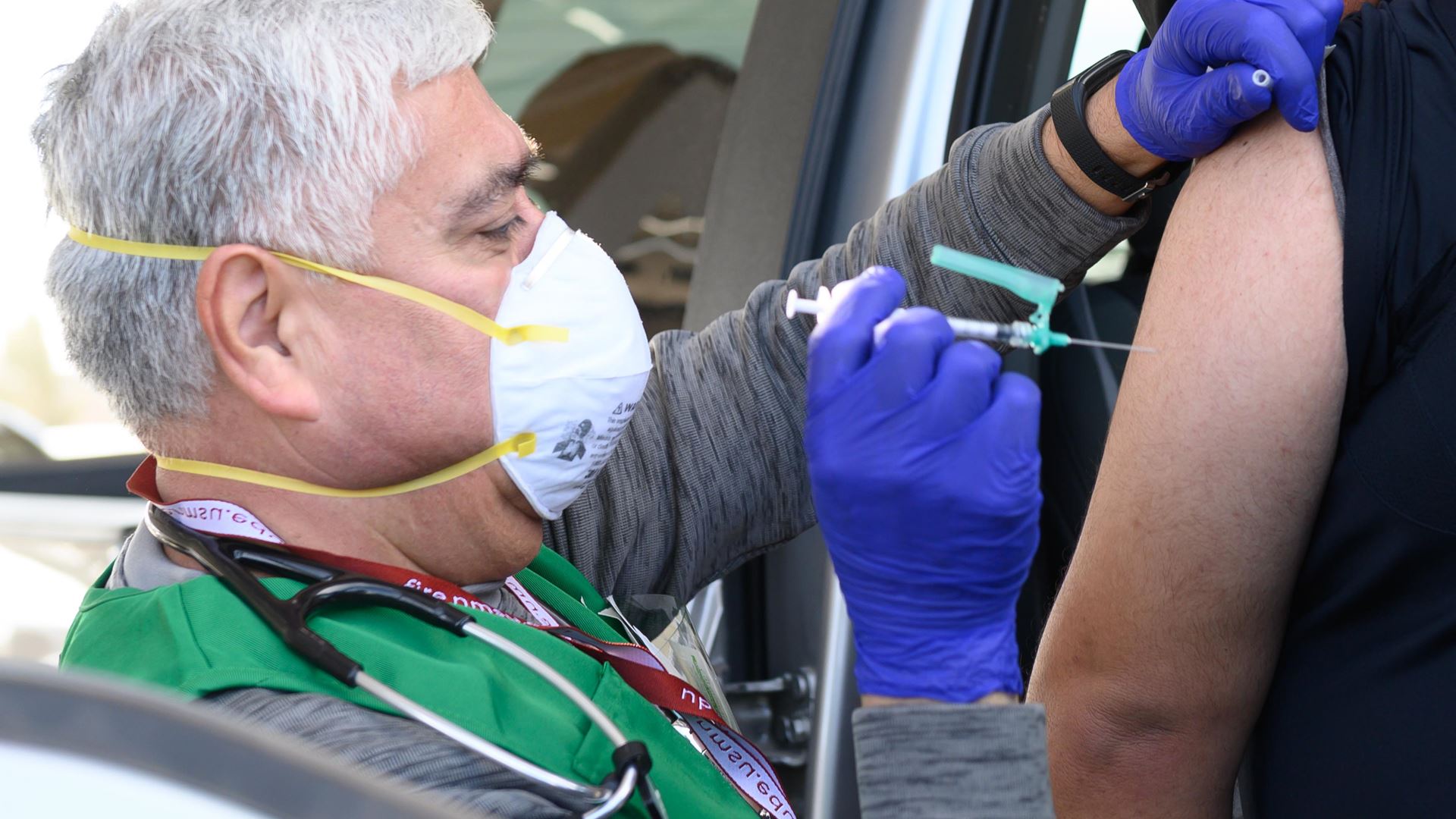
New Mexico State University distributed the Johnson and Johnson vaccine during the vaccination event Friday, April 9. The New Mexico Department of Health on Tuesday, April 13 announced that Johnson & Johnson’s COVID-19 vaccine distribution will be paused following the Centers for Disease Control and Prevention and Food and Drug Administration recommendations. This pause is out of an abundance of caution following six reports of “rare and severe” blood clots out of 6.8 million doses given among women between the ages of 18 and 48.
For those who have already received J&J vaccine, symptoms have occurred six to 13 days after vaccination and consist of severe headache, blurred vision, seizure, abdominal pain, leg pain or shortness of breath. Please note: these adverse events are extremely rare – less than one per million.
NMDOH advises those who have experienced any of these symptoms within three weeks after vaccination should contact their personal health care provider.